
IPCA is a popular Indian pharma organization and manufactures over 350 formulations and 80 APIs for various therapeutic segments. IPCA Hiring For Multiple Position.
For more than 60 years, IPCA has been partnering healthcare globally in over one hundred and twenty countries and in markets as diverse as Australia, Europe, Africa, Asia, and the US.
Over 20 years, IPCA has been playing a great role in the Indian APIs market, both in the anti-malarial and anti-hypertensive therapeutic segments. IPCA is the first manufacturer in India for APIs like Atenolol, Hydroxycholoquine Sulphate, Morantel Citrate, Pyrantel Pamoate, and Zaltoprofen.
- Basic Principle and Procedure of Fungal Staining
- What are Oxygen absorbers and How works?
- What is Pasteurization?
- What is Disinfectant and antiseptic? Disinfectants vs antiseptic
- Why More Men are Dying from Coronavirus than Women?
IPCA is one of the world’s largest manufacturers and suppliers of over a dozen APIs. These API are produced from the basic stage at manufacturing units inspected by the drug regulatory authorities like US-FDA, EDQM-Europe, WHO-Geneva, UK-MHRA, and many more. At IPCA, Quality Assurance is a human attitude of seeking sustainable betterment, in every aspect of our work. IPCA invests in constantly reinventing human quality through continuous training, exposure, and goal setting. In improving products, improve processes; and to do so.
IPCA has been awarded by Great Place to Work® – India in joint collaboration with The Economic Times as ‘Among the 100 Best Companies to Work in India 2010’ in a study conducted.
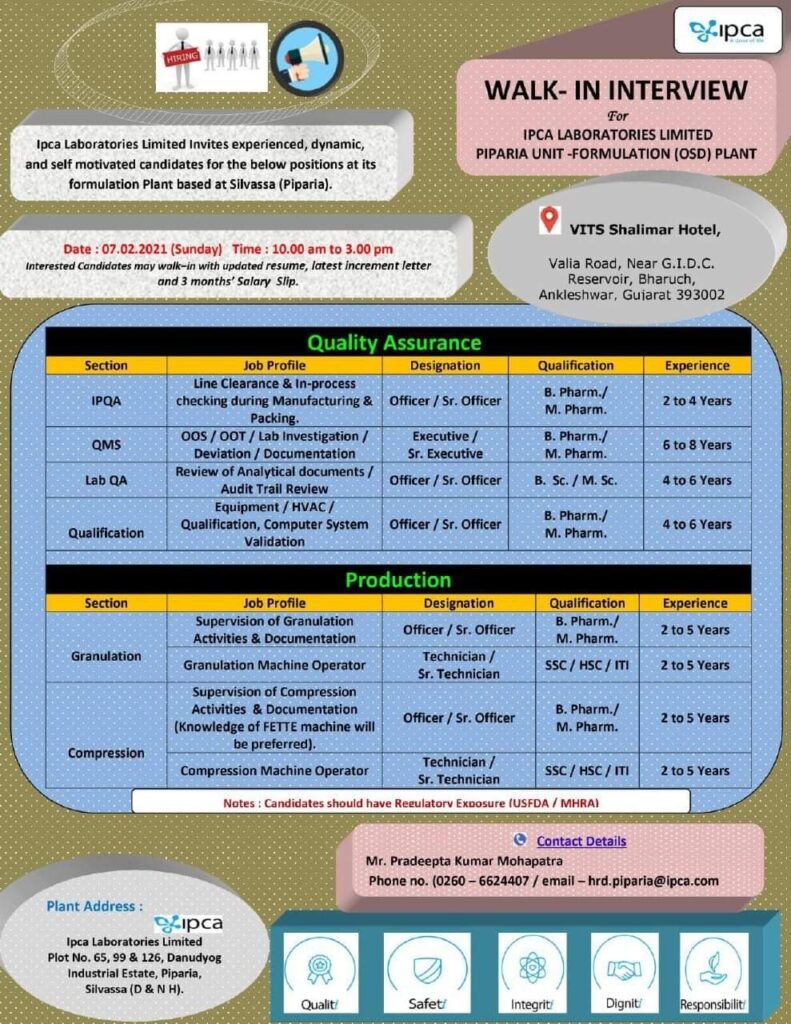
IPCA Hiring For Multiple Position for QA, IPQA, QMS, Lab QA, and OSD Production (B.SC/M.SC/ B.Pharm/ M.Pharm)
Interested candidates may search here to view Vacancies or apply at hrd.mumbai@ipca.com / hrdtech.mumbai@ipca.com
- What are Windows Security Policies or group policy management for the GxP computerized system?
- Clininvent Research is hiring Production and QC Chemist
- How to write User Requirement Specification?
- Chemex Global is Hiring Quality Head
- Bengal Beverages is hiring a SENIOR MAINTENANCE ENGINEER
- Ransh Pharma: Best Intermediates Manufacturing Unit at Visakhapatnam
- Ind-Swift-Laboratories Ltd (API Unit) is hiring for the R&D department
- Syngene International Ltd is Hiring M.Sc, M.Tech, M.Pharma
- Urgent Opening for Business Development Executive/ Manager
- Meyer Organics is Hiring for Quality Control, Production, and Store Departments.
- Torrent Pharma is hiring for the R&d Center Gandhinagar
- Amneal Walk in Interview for Ahmedabad Unit
- Endo India Par Formulation is Hiring for Quality Control Department
- QC Chemist
- Urgent Opening for Sales & Marketing Executive
- What is 3D Bioprinting? Basic Principles, Techniques, and Application of 3D Bioprinting.
- Basic Principle and Procedure of Fungal Staining
- OXYGEN MANAGEMENT FOR SUSTAINABLE ENVIRONMENT
- Urgent opening for Chemist for Food Lab and Metal Lab
- Urgent opening for Business Development department Manager for CRO.
- Urgent Requirement for Field Sales & Marketing Executive
- Urgent Opening for Plant Head- Pharmaceutical
- Cipla Walk-In-Interview at For multiple positions

Dear sir/mam
I am Manoj I have 5 year experience in regulatory industry as microbiologist
Thanks
Having 17 years pharmaceutical sales with 5 years manmangment postion as regional head covering noth and east India with diversified customers like gastro ,nephrologist,cardio,critical care,diabeto,
Quality control. RM sampling technical assistant 10th / 65. %+ 12th pass 50%
Experience 06 mounth QC lab sampling
Macleods pharmaceutical SEZ indor