
Aurobindo Pharma Ltd. is a reputed pharmaceutical company that delivers innovative solutions. Aurobindo Pharma is the market leader in Semi-Synthetic Penicillins, it has a presence in key therapeutic segments such as neurosciences, antiretrovirals, anti-diabetics cardiovascular, gastroenterology, and cephalosporins. Aurobindo has more than 300 products in various therapeutic segments. Aurobindo Pharma Walk-In-Interview For Multiple Position.
Aurobindo Pharma has successfully launched a range of affordable products that are accessible across the world. Aurobindo Pharma has fifteen state-of-the-art formulation production units located in India, the USA, Portugal & Brazil. Aurobindo facilities have received accreditation from the following regulatory bodies:
- US FDA (United States Food and Drug Administration)
- UK’s MHRA (United Kingdom’s Medicines and Health products Regulatory Agency)
- TGA Australia (Therapeutic Goods Administration)
- MCC South Africa (Medicines Control Council)
- ANVISA Brazil (National Health Surveillance Agency)
- Health Canada
- WHO (World Health Organization)
- GCC DR (Gulf Central Committee for Drug Registration)
- Basic Principle and Procedure of Fungal Staining
- What are Oxygen absorbers and How works?
- What is Pasteurization?
- What is Disinfectant and antiseptic? Disinfectants vs antiseptic
- Why More Men are Dying from Coronavirus than Women?
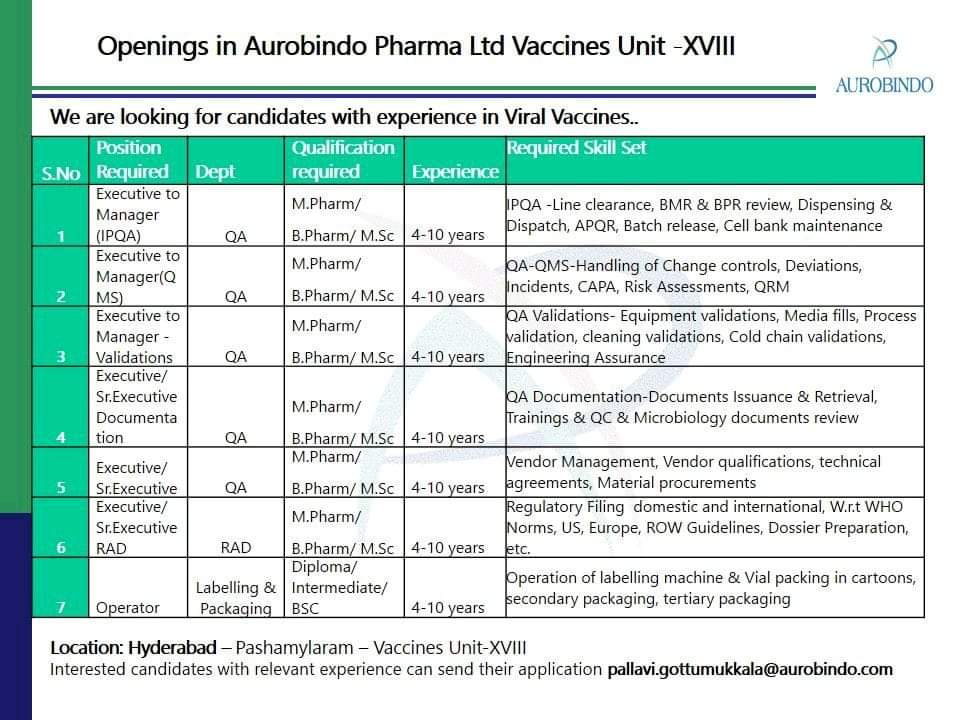
Aurobindo Pharma Walk-In-Interview at Hyderabad For IPQA, QA, RAD, Packing (M.Pharm, M.Sc, B.Pharm, D.Pharm, ITI, B.Sc)
For Job, Applications Send Your Resume to hr@aurobindo.com
Read Articles For Crack Interview
- What are Windows Security Policies or group policy management for the GxP computerized system?
- How to write User Requirement Specification?
- Ransh Pharma: Best Intermediates Manufacturing Unit at Visakhapatnam
- What is 3D Bioprinting? Basic Principles, Techniques, and Application of 3D Bioprinting.
- Basic Principle and Procedure of Fungal Staining
Aurobindo Pharma Key Formulation Facilities (8 in India, 1 each in US and Brazil)
Unit 3
- A multi-product oral dosage (OSD) form facility for non-cephalosporins and non-beta-lactams
Unit 4
- A dedicated manufacturing facility for generic sterile injectable (lyophilized and powder injections, prefilled syringes), ophthalmic and low volume parenteral.
Unit 7
- An ultra-modern unit based on the suite manufacturing concept delivering huge capacity.
- Manufactures non penicillins, non cephalosporins & ARVs
Unit 10
- Manufactures non-penicillins & non-cephalosporins products.
Unit 12
- A dedicated facility manufacturing oral and sterile beta-lactam formulations.
Unit 15
- An exclusive multi-product oral dosage form facility for supplying European markets.
- The unit has an installed annual capacity of 6 billion tablets and capsules.
Auronext
- Produces sterile penne formulations.
- Also manufactures sterile APIs.
Aurolife (Located in the US)
- USFDA approved cGMP compliant manufacturing facility located in New Jersey
- Manufactures non-penicillin and non-cephalosporin products
Brazil
- Manufacturing facility for SSPs, which caters to the requirements of Latin America
Aurobindo Pharma Regulatory History
Aurobindo Pharma has a very strong track record in regulatory filings. Aurobindo has applied for 150 US DMFs and about 2000+ filings in other countries. Aurobindo has close to 500 patents in place.
Find More Jobs at Aurobindo Pharma
- What are Windows Security Policies or group policy management for the GxP computerized system?
- Clininvent Research is hiring Production and QC Chemist
- How to write User Requirement Specification?
- Chemex Global is Hiring Quality Head
- Bengal Beverages is hiring a SENIOR MAINTENANCE ENGINEER
- Ransh Pharma: Best Intermediates Manufacturing Unit at Visakhapatnam
- Ind-Swift-Laboratories Ltd (API Unit) is hiring for the R&D department
- Syngene International Ltd is Hiring M.Sc, M.Tech, M.Pharma
- Urgent Opening for Business Development Executive/ Manager
- Meyer Organics is Hiring for Quality Control, Production, and Store Departments.
- Torrent Pharma is hiring for the R&d Center Gandhinagar
- Amneal Walk in Interview for Ahmedabad Unit
- Endo India Par Formulation is Hiring for Quality Control Department
- QC Chemist
- Urgent Opening for Sales & Marketing Executive
- What is 3D Bioprinting? Basic Principles, Techniques, and Application of 3D Bioprinting.
- Basic Principle and Procedure of Fungal Staining
- OXYGEN MANAGEMENT FOR SUSTAINABLE ENVIRONMENT


I am a Pharma person and interested. thanks, Pharma Awareness
I am M.pharma student and interested to join job at Aurobindo Pharma
Presently working as a RBM of a pharma Company….total experience of 20 years …with a good team
Comments are closed.