
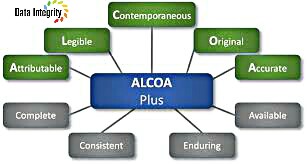
Data integrity is key in a pharmaceutical quality system ensuring that products are of the desired quality. Data Integrity is increasingly becoming a critical GMP issue in the Pharmaceutical and Medical Device Industry. ALCOA-plus. A normally used word form for “attributable, legible, contemporaneous, original and accurate”
The descriptor ALCOA has been around since the 1990’s ensuring by regulated industries as a framework for guaranteeing data integrity, and good Documentation practice (GDP). Originally the ALCOA descriptor was used as a straightforward and practical guide by Stan Woolen of us Food and Drug Administration on the North American nation Food and Drug Administration expectations relating to proof of each paper-based, electronic, and hybrid quality systems or records. it’s currently a GMP necessary that each one employee ought to be trained in ALCOA and data Integrity to confirm a corporate culture that values data integrity.
- What are Windows Security Policies or group policy management for the GxP computerized system?
- How to write User Requirement Specification?
- Ransh Pharma: Best Intermediates Manufacturing Unit at Visakhapatnam
- What is 3D Bioprinting? Basic Principles, Techniques, and Application of 3D Bioprinting.
- Basic Principle and Procedure of Fungal Staining
Currently, ALCOA has been expanded on to ALCOA Plus (currently used by the FDA, WHO, PIC/S and GAMP – Data Integrity), this neatly requires all data to have the following qualities:
| Term | Defination |
| Attribute | Who aquire the data or perfofmed an action and when? |
| Legible | Can you read the data and any entries? |
| Contemporeneous | Data are recorded at the time they are generated or observed |
| Original | A written Printout or observed or certified copy there of |
| Accurate | The term accurate means data are correct, truthful, complete, valid and reliable |
The Newer Alcoa Plus adds a few attribute to the list
| Term | Defination |
| Complete | All the Data including any repeat or reanalysis performed on the sample, nothing has been deteled (Evidence: Audit trial). |
| Consistent | All elements of the analysis such as the sequence of events follow on and are date or time stamped in the expected sequence. |
| Enduring | Not Recorded on the back of envelops, cigarette packets, sticky notes or the sleeves of a coat but in notrbooks or electronic media in the data systems of instruments |
| Available | Can be accessed for review and audit or inspection over the lifetime of the record |
Why Has Data Integrity Become a Hot Topic?
Inspectors from regulative bodies inspected the Pharmaceutical business, and that they found that the data wasn’t correct. If a pharmaceutical business produces medicine within the declared parameters, then it’s approved and available within the marketplace for patients. If inspectors realize that the data is changed, then the drug isn’t approved. meaning revenue loss for business and medicines not offered for patients.
Beginning in 2014 there was a marked increase in warning letters to Indian drug manufacturers addressing data integrity because the Food and Drug Administration uncovered issues throughout regulatory inspections.
- What are Windows Security Policies or group policy management for the GxP computerized system?
- Clininvent Research is hiring Production and QC Chemist
- How to write User Requirement Specification?
- Chemex Global is Hiring Quality Head
- Bengal Beverages is hiring a SENIOR MAINTENANCE ENGINEER
- Ransh Pharma: Best Intermediates Manufacturing Unit at Visakhapatnam
- Ind-Swift-Laboratories Ltd (API Unit) is hiring for the R&D department
- Syngene International Ltd is Hiring M.Sc, M.Tech, M.Pharma
- Urgent Opening for Business Development Executive/ Manager
- Meyer Organics is Hiring for Quality Control, Production, and Store Departments.
- Torrent Pharma is hiring for the R&d Center Gandhinagar
- Amneal Walk in Interview for Ahmedabad Unit
- Endo India Par Formulation is Hiring for Quality Control Department
- QC Chemist
- Urgent Opening for Sales & Marketing Executive
- What is 3D Bioprinting? Basic Principles, Techniques, and Application of 3D Bioprinting.
- Basic Principle and Procedure of Fungal Staining
- OXYGEN MANAGEMENT FOR SUSTAINABLE ENVIRONMENT
- Urgent opening for Chemist for Food Lab and Metal Lab
- Urgent opening for Business Development department Manager for CRO.
- Urgent Requirement for Field Sales & Marketing Executive
- Urgent Opening for Plant Head- Pharmaceutical

Very useful topic for current days.
Thanks for sharing current topic.
Well describe
Thanks
Good information
This is Yogesh Ghule, Looking after Quality Control,
in Zydus Healthcare Ltd.
Comments are closed.